Combine the structures of IT documentation with the advantages of a CMDB. The abbreviation CMDB stands for Configuration Management Database. A database that is used to record your technical documentation in detail, from hardware to software to network (configuration items). As a Premium Partner, Certified Support Partner and Development Partner, we recommend the 360° IT documentation software i-doit with a scope that goes far beyond the idea of a CMDB. For example, to manage the vehicle fleet, to fulfill data protection according to DSGVO or to implement industry-specific safety standards. An extension to document medical technology is therefore only logical. In this article, we will give you 4 significant reasons to document medical products with the i-doit Add-on MedTec speak.
Medical technology documentation with i-doit – Why does it make sense?
The many advantages that i-doit offers as a configuration management database solution for IT can also be used by the medical technology sector. Instead of maintaining data in manual form or in a spreadsheet program, i-doit not only offers a better graphical interface, but also the ability to display, easily document and evaluate links or relationships to people, contracts or other assets. Access is possible via web access. The installation on the server side is usually used “on-premise”, which means that all data is located on your server. Alternatively, there is also the possibility to use i-doit in the cloud.
Documenting according to medical technology operator regulation
The MedTec add-on gives you the option of documenting your medical technology in accordance with the requirements of the Medical Device Operator Ordinance (MPBetreibV). For each medical device asset, you can access the medical device book, which is required by §12 of the standard. An inventory according to §13 MPBetreibV is also included and lists all documented medical devices with the relevant attributes. Deadlines required by law, such as safety and metrological inspections, can be planned and documented in the same way as maintenance.
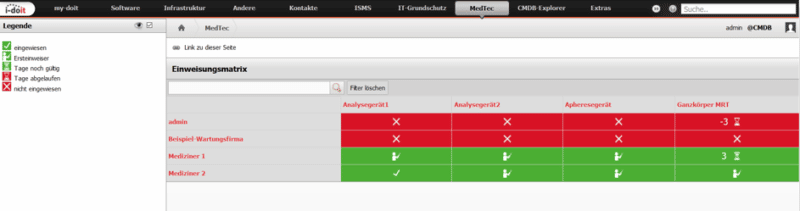
In §4 of the MPBetreibV, instruction documentation is required. The MedTec Add-on helps you list your staff as rows and your devices as columns through an interactive briefing matrix. There are filter options for both people and devices, so that the view can be narrowed down to a specific department, for example. However, the planning or creation of a new briefing is not object-related but model-related. Evidence of briefings carried out can be stored in an audit-proof manner. From the matrix, it is also possible to see directly whether an admission is about to expire, has recently expired, whether someone is a “first-time admitter” or whether no admission has yet taken place at all.
Furthermore, you can assign the associated risk class to the medical devices and assign a contact assignment with the role “Responsible person according to §5 MPG“.
Of course, the documentation of malfunctions must not be missing. The integration of a ticket system, from which these messages can be imported, is particularly suitable for this purpose. On the other hand, it is possible to define spare parts for the devices for which an ordering process can be implemented in a ticket system.
Extensibility and interfaces – a strength of our open source solution
In addition to the additional features provided by the MedTec add-on, you can strongly individualize the software and adapt it to your needs. Be it creating your own fields and config item classes for the CMDB, using preconfigured interfaces like a synchronization from the Active Directory or creating your own interfaces using the well documented JSON RPC API interface. i-doit also offers a wide range of integration options as standard, allowing you to document your CMDB fully automatically. In the infrastructure area, for example, there are interfaces to OCS Inventory, JDisc Discovery and CheckMK (see further add-ons). There are also native interfaces and add-ons for various ticket systems. This allows, for example, reported malfunctions to be displayed directly on the medical devices in i-doit or processes such as spare parts orders to be linked to the respective Config items.
Competitive pricing model
CAFM software specializing in medical technology is usually very price-intensive. We can also score on this front with our open source solutions. The pricing model of the i-doit CMDB solution is transparent and dependent on the number of config items to be documented.
Have we aroused your interest? Please contact us! We are very happy to develop and implement individual solutions with you.
MedTec – News from the medical technology add-on | Live demo of the new functions such as the referral matrix
premium partner
becon is i-doit Premium Partner,Certified Support Partner and Development Partner of synetics GmbH and thus provides the all-round carefree package.



Subscribe to our blog!
We’re not just going to talk about us here,
but above all provide interesting information about the change of IT with all its technological facets.
360° IT Documentation
i-doit Professional – An ISMS with the advantages of a generic CMDB
Fact Sheet
More than just IT documentation
i-doit is more than just the best open source based platform for IT documentation. With an intelligent relationship model, the ability to model IT services, manage configurations to devices and applications, and visualize dependencies and statistics, i-doit becomes a business-ready CMDB. In this fact sheet we describe the benefits of i-doit and the service packages, add-ons and service packages becon offers you.
blog
Related blog articles
Decision support in IT management: Interview with the experts from DATAGERRY
Which CMDB tool is right for your company? We spoke to the experts behind DATAGERRY. Jakob Semere, Head of Applications at becon GmbH, and Kai Schubert-Altmann, Senior Consultant and Web Application Manager at becon GmbH, share their experiences with CMDB & Asset Management.
DATAGERRY | Release 2.0
We are pleased to announce the new version 2.0 of DATAGERRY! It is now available to download. The update brings new functions such as the DATAGERRY Assistant and the “Locations” feature.
becon is i-doit Best Performing Partner 2022
becon GmbH was awarded “i-doit Best Performing Partner 2022” by synetics GmbH. This award is a recognition of our continued efforts…
Contact
Instant contact
Do you have any questions, suggestions, requests or are you facing a particular challenge? We look forward to hearing from you!
+49 (0) 89 608668-0





